Corrective action preventive action8/28/2023  The site personnel signature/delegation form was not completed correctly or in a timely manner. Delegated responsibilities were assigned to everyone regardless of their role on the study.The auditor reviews the site documentation including personnel signature/delegation forms and source documents and notes the following issues: 
Case StudyĪn auditor is at an investigator’s site performing a clinical trial audit. Clinical research professionals are often unable to give their full attention to every detail of the study due to the speed in which the trial progresses, the strain on company resources, or many of the other day-to-day factors that impact ones’ time and attention. Today, clinical trial professionals are provided with guidelines, regulations, company policies, procedures, and endless amounts of training to help them with the conduct of clinical trials.ĭespite all the training one receives or all the guidelines, regulations, company policies, and procedures in place to assist with the conduct of a clinical-trial, problems including unanticipated situations or safety issues may arise (Table 1). 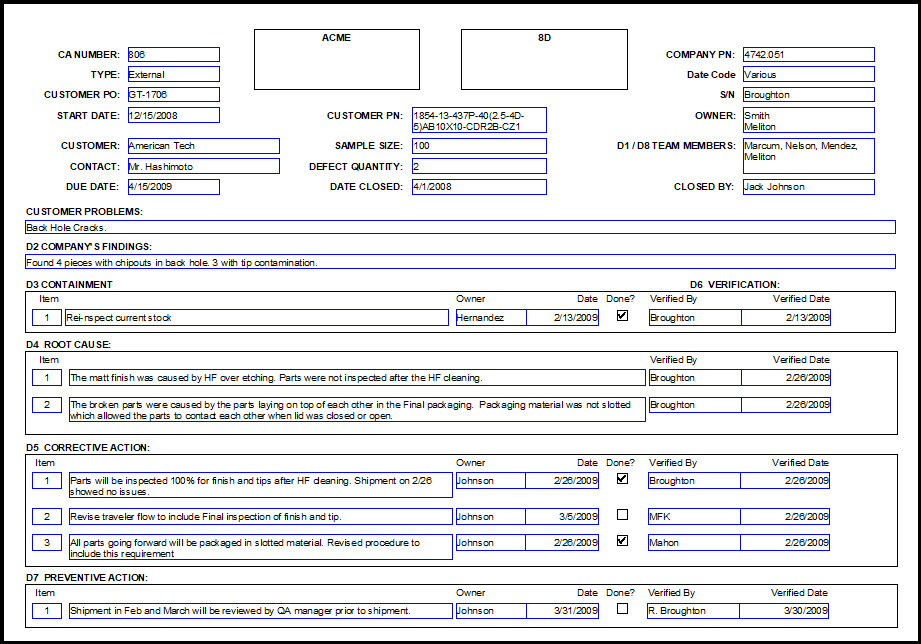 
Actual case study illustrates some of the common problems in clinical trials. This article provides an overview of the root cause of these problems and how to ensure that corrective and preventive actions are addressing the actual problem rather than its symptoms. Senior Manager, Clinical Compliance, MedImmuneĬhartered MCIPD, Senior Manager, Scientific & Compliance Training, MedImmuneĪbstract : Clinical trial findings from audits reveal the same type of problems year after year despite the implementation of quality systems, compliance training, and corrective and preventive action plans. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
